A New Gold Standard for Stool Sample Storage
Ensuring Capture of the True Microbiome Profile
As researchers and clinicians delve into the gut with a multi-omics approach, multiple aspects of the gut microbiome are considered. For example, the microbial genome, transcriptome, and metabolites for the entire microbial community are profiled. This helps shed a light on the composition (genome) and activity (transcriptome and metabolome) within the gut microbiome. Stool samples are often used to gain a representative snapshot of the gut microbiome, and as we learn more about this sample type, we strive to ensure that they are handled with best practices in mind.
Since stool samples are often used as a proxy for gut microbiome studies, the collection and storage method selected must halt microbial activity. Bacteria are responsive to their external environment, so once the sample is exposed to conditions outside of the body, the microbiome profile will experience a shift due to this adaptation. The current gold standard for stool sample preservation is freezing, but as we reviewed in our past blog (Revisiting the Gold Standard for Stool Sample Storage), freezing of the sample will lead to the lysis of Gram-negative bacteria and subsequent exposure of their nucleic acids to nucleases, whereas chemical preservation lyses all bacteria and inactivates the nucleases in stool samples.1,2
NORBLOG
Want to hear more from Norgen?
Join over 10,000 scientists, bioinformaticians, and researchers who receive our exclusive deals, industry updates, and more, directly to their inbox.
For a limited time, subscribe and SAVE 10% on your next purchase!
SIGN UP
A larger comparison by Plauzolles et al. (2022) evaluated the technical aspects of several collection and storage methods, including freezing, chemical preservation, room temperature storage, and fecal swabs. DNA was purified and the 16S rRNA gene was subsequently sequenced from these samples.3 The authors identified Norgen's Stool Nucleic Acid Collection and Preservation Tubes as the best performing solution for the stabilization of the gut microbiome. In Figure 1, Norgen was found to be most similar to the frozen samples, being closest to the reference (bottom of the graph).
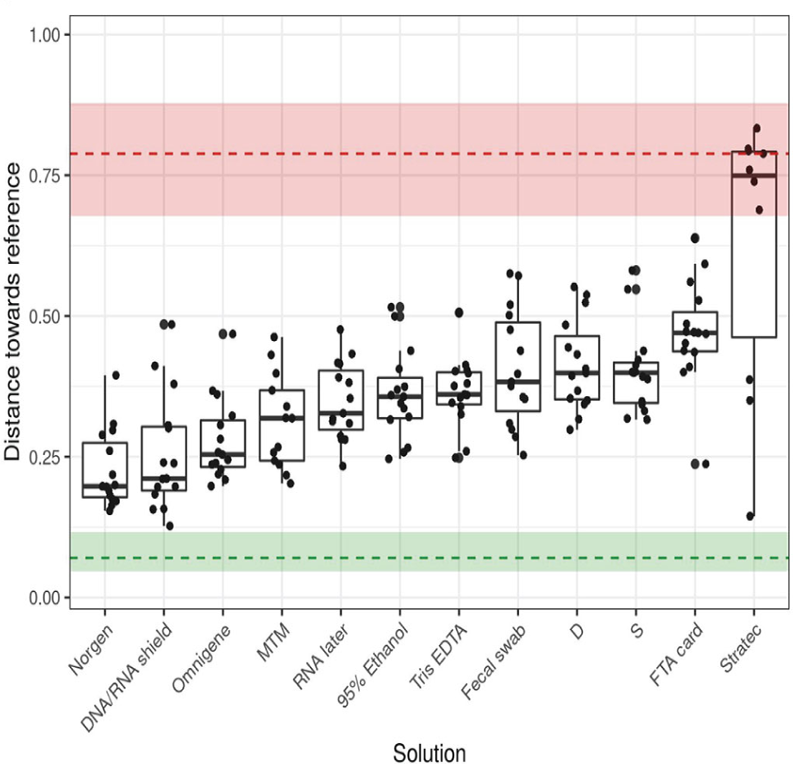
This recent comparison and previous results show that freezing stool samples lyses Gram-negative bacteria, which leads to the exposure of Gram-negative bacterial DNA to nucleases. Although freezing is known as the current gold standard, the true microbial profile may be captured more accurately through Norgen’s chemical preservation method. To learn more about stool collection and preservation, watch our on-demand webinar, “A Multi-Omics Look Into the Stool Microbiome".
-
Walker VK, Palmer GR, Voordouw G. Freeze-Thaw Tolerance and Clues to the Winter Survival of a Soil Community. Applied and Environmental Microbiology. 2006;72(3):1784-1792. doi:10.1038/s41598-019-53183-5
-
Watson EJ, Giles J, Scherer BL, Blatchford P. Human faecal collection methods demonstrate a bias in microbiome composition by cell wall structure. Scientific Reports,2019;9(1):1-8. doi:10.1038/s41598-019-53183-5
-
Plauzolles A, Toumi E, Bonnet M, et al. Human Stool Preservation Impacts Taxonomic Profiles in 16S Metagenomics Studies. Front Cell Infect Microbiol ,2022;12:722886. doi:10.3389/fcimb.2022.722886