Complete Saliva Workflow
Saliva Collection and Preservation Devices
Best-in-class solutions for the rapid collection, and preservation of saliva RNA and DNA
Featured Publication:
Pre-analytical sample stabilization by different sampling devices for PCR-based COVID-19 diagnostics.
Explore the findings
Please select the type of Saliva Tube you are interested in
Saliva Collection and Preservation Devices
Select each molecule to learn more about Norgen's unique tube and how it separates DNA/RNA from saliva samples
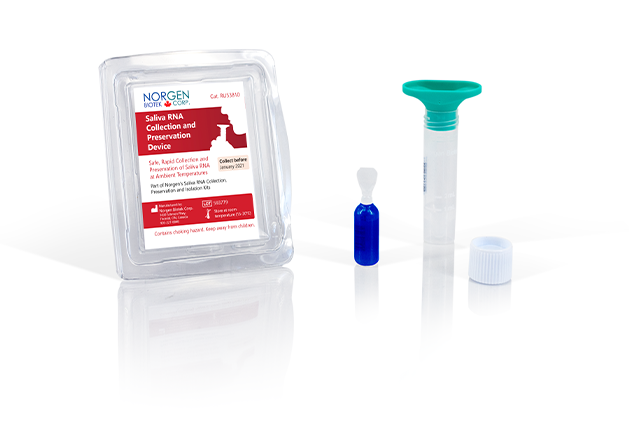
Saliva RNA Collection and Preservation Devices
 RNA×
RNA×
 Bacteria×
Bacteria×
 Viruses×
Viruses×
Saliva RNA Collection and Preservation Devices
Norgen’s Saliva RNA Collection and Preservation Devices are designed for 1) simple and non-invasive saliva collection and 2) preservation of RNA in saliva samples at ambient temperature. Saliva samples are collected by spitting inside the Collection Funnel which has been assembled with the Collection Tube. After collecting the required volume of saliva the Collection Funnel is removed and the contents of the Preservative Ampoule are then added and mixed with the collected saliva. The Saliva Collection Tube is subsequently sent to the laboratory for RNA isolation and analysis. The Saliva RNA in preserved samples is stable for up to 2 months at room temperature. This kit is ideal for collecting and preserving RNA samples for epidemiological and population studies.


RNA
Saliva is a source of both cellular and cell-free host RNA as well as exogenous RNA from bacterial or viral cells. The majority of host cellular RNA originates from the buccal mucosa and can be used for transcriptomic analyses to identify which functional pathways are active within the cells. Host cell-free RNA in the saliva is typically introduced into the saliva via filtration from the bloodstream into the salivary glands. While RNA is usually degraded by nucleases in the bloodstream, fragments that are bound to proteins (i.e. ribosomes and other proteins) or encapsulated inside exosomes are protected from degradation and remain in circulation. Once purified, cell-free RNA can be analyzed and used as a biomarker for various diseases and conditions.
Links
Bacteria
Bacteria in saliva are part of the oral microbiome which consists of approximately 700 different species that exist in a symbiotic relationship with the host. A disruption in this relationship, known as dysbiosis, can lead to various oral diseases or systemic diseases. Furthermore, oral dysbiosis is also associated with certain head and neck cancer development. Therefore, identifying changes in salivary bacterial composition can not only be used to diagnose certain oral or systemic diseases but also to understand the role it plays in the alteration of functional and metabolic pathways associated with the development of certain head and neck cancers.
Viruses
Saliva can not only contain viable intact viruses but also viral nucleic acids that can be used to screen for several diseases. Examples of such viruses include Influenza virus A, SARS-CoV-2, Herpes simplex virus 1 and 2, Rabies virus, Epstein-Barr virus, Cytomegalovirus, Rhinoviruses, Hepatitis A, B & C viruses, Measles virus, and Human papillomavirus. Being non-invasive and easy to collect, saliva, therefore, is a valuable specimen to detect viral infections.
Links
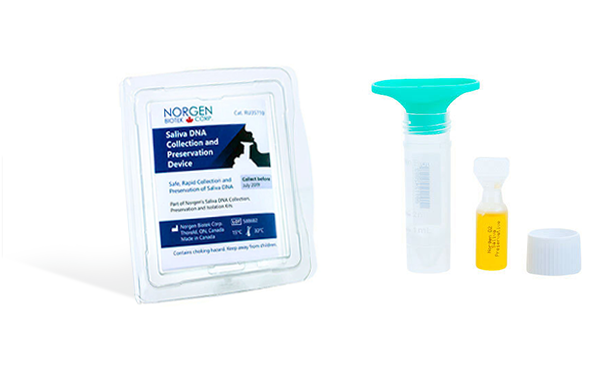
Saliva DNA Collection and Preservation Devices
 Cellular g-DNA×
Cellular g-DNA×
 Bacteria×
Bacteria×
 Viruses×
Viruses×
Saliva DNA Collection and Preservation Devices
Norgen's Saliva DNA Collection and Preservation Devices are designed for 1) simple and non-invasive saliva collection and 2) preservation of DNA in saliva samples at ambient temperature. Saliva samples are collected by spitting inside the Collection Funnel which has been assembled with the Collection Tube. After collecting the required volume of saliva the Collection Funnel is removed and the contents of the Preservative Ampoule are then added and mixed with the collected saliva. The Saliva Collection Tube is subsequently sent to the laboratory for DNA isolation and analysis. DNA can be isolated from the preserved saliva samples using Norgen's Saliva DNA Isolation Kit (Cat# RU45400). Each of Norgen's Collection Tubes is labeled with a unique serial number that can be used for secure and anonymous tracking of the sample. The saliva DNA in preserved samples is stable for more than 2 years at room temperature. This kit is ideal for collecting and preserving DNA samples for epidemiological and population studies.

Cellular g-DNA
The majority of DNA in saliva originates from the buccal epithelial cells shed from the inner lining of the mouth and white blood cells. Also present in the saliva is cell-free DNA that originates from the apoptosis or necrosis of host cells and is usually present in very low concentrations. A clinically important type of cell-free DNA is circulating tumor DNA or ct-DNA which originates from tumor cells. By examining the mutations within the ct-DNA, or its methylation profile, ct-DNA can act as a key biomarker for the diagnosis and monitoring of cancer development. Another important source of DNA in the saliva is bacterial cells inhabiting the oral cavity. This DNA can not only be used to profile the salivary microbiome but is also important in detecting pathogens that cause oral diseases.
Links
Bacteria
Bacteria in saliva are part of the oral microbiome which consists of approximately 700 different species that exist in a symbiotic relationship with the host. A disruption in this relationship, known as dysbiosis, can lead to various oral diseases or systemic diseases. Furthermore, oral dysbiosis is also associated with certain head and neck cancer development. Therefore, identifying changes in salivary bacterial composition can not only be used to diagnose certain oral or systemic diseases but also to understand the role it plays in the alteration of functional and metabolic pathways associated with the development of certain head and neck cancers.
Links
Viruses
Saliva can not only contain viable intact viruses but also viral nucleic acids that can be used to screen for several diseases. Examples of such viruses include Influenza virus A, SARS-CoV-2, Herpes simplex virus 1 and 2, Rabies virus, Epstein-Barr virus, Cytomegalovirus, Rhinoviruses, Hepatitis A, B & C viruses, Measles virus, and Human papillomavirus. Being non-invasive and easy to collect, saliva, therefore, is a valuable specimen to detect viral infections.
Features & Benefits
- Preserved RNA is stable for up to 2 months at room temperature
- Sample collection and preservation in one convenient kit
- Samples are non-infectious and can be handled and shipped safely at ambient temperature
- Non-invasive, user-friendly collection and suitable for self collection
- Hassle-free and cost/effective - no cold shipping/storage needed
- Compatible with RNA isolation methods
- Preserve high-quality RNA that is compatible with any downstream application
- CE-IVD marked version available
- Preserved DNA is stable for 2 years at ambient temperature
- Non-invasive, user-friendly collection and suitable for self collection
- Sample collection and preservation in one convenient kit
- Samples are non-infectious and can be handled and shipped safely at ambient temperature
- Hassle-free and cost/effective - no cold shipping/storage needed
- Isolate high-quality nucleic acids for any downstream applications
- CE-IVD marked version available
Supporting Data
Figure 1. Stability of RNA Preserved in Norgen's Saliva RNA Preservative at room temperature for up to 2 months. Saliva samples were collected from numerous donors and mixed, and then an equal volume of Norgen's Saliva RNA Preservative was added to the saliva. The preserved saliva was then stored at room temperature for up to 2 months. Saliva RNA was subsequently isolated at day 0, day 4, day 6, day 16, 1 month and 2 months from 0.25 mL of the saliva/preservative sample using the supplementary protocol associated with Norgen's Total RNA Purification Kit (Cat. 17200). For visual analysis, 7.5 µL from 50 µL elution was mixed with RNA loading dye and was run on a1x MOPS agarose gel. RNA integrity was maintained after the saliva samples werestored for up to 2 months at ambient temperatures in Norgens Saliva RNA Preservative.
Figure 2. Stability of three house keeping genes over a 2 month period. GAPDH, Human 18S rRNA and beta actin genes were monitored from saliva samples stored at ambient temperature in Norgen's Saliva RNA Preservative. RNA was isolated from 250 µL of preserved samples on day 0, day 4, day 6, day 10, day 16, day 30 and day 60 using Norgen's Total RNA Purification Kit (Cat. 17200). Next, 4 µL of elution was used in 20 µL of the real-time PCR reaction (SYBR Green I) using CFX96 TouchTM Real-Time PCR Detection System (Bio-Rad). All of the genes were preserved and stable over 2 month period.
Figure 3. Norgen's Saliva RNA Preservative is able to successfully preserve saliva RNA samples when used outside of the ideal range of 1:1 at room temperature for 3 days. As the amount of saliva added to 2 mL of Saliva RNA Preservative increased from 0.2 mL to 3 mL, RNase protein and β-actin gene were monitored for RNA stability using one-step RT-PCR. The results indicated that the variation of the saliva input volumes did not significantly influence the RNA gene detection.
Figure 4. Detection of Viral, Bacterial and Human RNA from Preserved Saliva Samples. Saliva (2 mL) collected from a single healthy donor was mixed with 2 mL of Saliva RNA Preservative. Norgens COVID-19 positive control from the 2019-nCoV TaqMan RT-PCR Kit (E/RdRP gene) (Cat. TM67200) was spiked into the preserved saliva along with HeLa cells (1.6 m cells /ml ) to generate a contrived sample. From the contrived sample, total RNA was purified using Norgens Total RNA Purification Kit (Cat. 17200) and 5 genes (16s rRNA, COVID-19 E gene, RdRP-P1 and P2 gene and RP) were analyzed by PCR or RT-PCR to detect viral, bacterial and human genes.
Supporting Data
Figure 1. Stability of DNA Preserved in Norgen's Saliva DNA Preservative at Room Temperature for over 2 Years. Saliva samples were collected from numerous donors and mixed, and then an equal volume of Norgen's Saliva DNA Preservative was added to the saliva. The preserved saliva was then stored at room temperature for up to 24 months. Saliva DNA was subsequently isolated at 4 months, 8 months, 16 months and 24 months from 0.5 mL of the saliva/preservative sample using Norgen's Saliva DNA Isolation Kit (Cat. RU45400). For visual analysis, 100 µL of the purified DNA was run on an agarose TAE gel. As it can be seen, there is no evidence of DNA degradation after the saliva samples are stored for 24 months at ambient temperatures in Norgen's Saliva DNA Preservative. Furthermore, the size of the DNA was maintained over 24 kb for the entire 24 month period. M: Norgen's UltraRanger 1 Kb DNA Ladder (Cat. 12100).
Figure 2. GAPDH Real Time PCR Amplification of Saliva DNA from Saliva Samples Stored at Room Temperature from Day 0 up to 24 Months. All isolated saliva DNA samples were successfully amplified without any sign of PCR inhibition and difference in Ct. Red: Day 0, Blue: 4 months, Purple: 8 months, Green: 16 months and Sky blue: 24 months. Black: No template control.
Figure 3. V3-V4 16s rRNA PCR amplification for Illumina MiSeq 16s rRNA library preparation. The saliva DNA was isolated using Norgen's Saliva DNA Isolation Kit (Cat. RU45400) from saliva that had been preserved for various periods of time up to 2 years at room temperature in Norgens Saliva DNA Preservative. The PCR result shows the clean PCR amplification at ~550 bp indicating the right target of V3-V4 16s rRNA , with no difference between day 0 and 24 months. M: FastRunner DNA Ladder (Cat. 12800)
Isolate High-Quality DNA and RNA from Saliva Samples
Use the interactive workflow to find the product right for you
Saliva
-
DNA
Samples collected with Norgen's Saliva DNA Collection and Preservation Device (49000, RU49000)
-
Saliva DNA Isolation Kits
Rapidly purify high-quality DNA from preserved and fresh saliva samples.
-
-
RNA
Samples collected with Norgen's Saliva RNA Collection and Preservation Device (53800, RU53800)
-
Saliva RNA Purification Kits
Rapidly purify total RNA from preserved and non-preserved saliva and nasal throat/swabs
-
-
Saliva DNA Purification Kits
Rapidly purify high-quality DNA from preserved and fresh saliva samples.
-
Saliva DNA Isolation Kit (Cat. RU45400)
Maximum Input Volume: 500 μL preserved saliva or 250 μL fresh saliva
-
Saliva DNA Isolation 96-Well Kit (Cat. RU35200)
Maximum Input Volume: 500 μL preserved saliva or 250 μL fresh saliva
-
Saliva DNA Isolation Kit Dx (Cat. Dx45400)
Maximum Input Volume: 500 μL preserved saliva or 250 μL fresh saliva
CE-IVDR marked
ideal for in vitro diagnostic use
-
Saliva DNA Isolation Kit (Magnetic Bead System) (Cat. RU55400)
Maximum Input Volume: 500 μL preserved saliva or 250 μL fresh saliva
-
Saliva DNA Isolation 96-Well Kit (Magnetic Bead System) (Cat. RU62900)
Maximum Input Volume: 500 μL preserved saliva or 250 μL fresh saliva
-
-
Saliva RNA Isolation Kits
Rapidly purify total RNA from preserved and non-preserved saliva and nasal throat/swabs
-
Saliva/Swab RNA Purification Kit (Cat. 69100)
Maximum Input Volume: 250 μL of fresh or preserved saliva
-
Saliva/Swab RNA Purification Kit Dx (Cat. Dx69100)
Maximum Input Volume: 250 μL of fresh or preserved saliva
CE-IVDR marked
ideal for in vitro diagnostic use
-
Saliva/Swab RNA Purification 96-Well Kit (Cat. 69300)
Maximum Input Volume: 250 μL of fresh or preserved saliva
-
Saliva/Swab RNA Purification 96-Well Kit Dx (Cat. Dx69300)
Maximum Input Volume: 250 μL of fresh or preserved saliva
CE-IVDR marked
ideal for in vitro diagnostic use
-
Saliva
-
DNA
Samples collected with Norgen's Saliva DNA Collection and Preservation Device (49000, RU49000).
-
Saliva DNA Purification Kits
Rapidly purify high-quality DNA from preserved and fresh saliva samples.
-
Saliva DNA Isolation Kit
Maximum Input Volume: 500 μL preserved saliva or 250 μL fresh saliva
-
Saliva DNA Isolation 96-Well Kit
Maximum Input Volume: 500 μL preserved saliva or 250 μL fresh saliva
-
Saliva DNA Isolation Kit Dx
Maximum Input Volume: 500 μL preserved saliva or 250 μL fresh saliva
CE-IVDR marked
ideal for in vitro diagnostic use
-
Saliva DNA Isolation Kit (Magnetic Bead System)
Maximum Input Volume: 500 μL preserved saliva or 250 μL fresh saliva
-
Saliva DNA Isolation 96-Well Kit (Magnetic Bead System)
Maximum Input Volume: 500 μL preserved saliva or 250 μL fresh saliva
-
-
-
RNA
Samples collected with Norgen's Saliva RNA Collection and Preservation Device (58300, RU58300)
-
Saliva RNA Isolation Kits
Rapidly purify total RNA from preserved and non-preserved saliva and nasal throat/swabs
-
Saliva/Swab RNA Purification Kit
Maximum Input Volume: 250 μL of fresh or preserved saliva
-
Saliva/Swab RNA Purification Kit Dx
Maximum Input Volume: 250 μL of fresh or preserved saliva
CE-IVDR marked
ideal for in vitro diagnostic use
-
Saliva/Swab RNA Purification 96-Well Kit
Maximum Input Volume: 250 μL of fresh or preserved saliva
-
Saliva/Swab RNA Purification 96-Well Kit Dx
Maximum Input Volume: 250 μL of fresh or preserved saliva
CE-IVDR marked
ideal for in vitro diagnostic use
-
-
Discover Norgen's NGS Services and Capture the Most From Your Samples
Next Generation Sequencing
Designed to Discover
- Illumina® MiSeq Sequencing Platform
- Fast Turnaround Time (3 - 4 weeks)
-
Small RNA (microRNA) Sequencing
- Capture a complete range of small RNA and miRNA species
- Sequence ultra-low inputs from liquid biopsies and exosomes
- Understand post-transcriptional regulation with bioinformatics analysis service
- Examine differential expression and discover novel biomarkers
-
SARS-CoV-2 Sequencing
- Sequence entire SARS-CoV-2 genome from saliva sample
- Detect novel SARS-CoV-2 variants using COVID-19 ARTIC Amplicon Sequencing Platform
- Examine inter-individual and intra-individual variations of the SARS-COV-2 virus
-
Metagenomics Sequencing
- Comprehensive service for 16S rRNA
- DNA extraction service included
- Analyze entire bacterial communities with comprehensive bioinformatics analysis
- Identify phylogenetic or taxonomic classifications
In our accredited state-of-the-art laboratory, we offer comprehensive Next-Generation Sequencing (NGS) services to provide full workflow solutions, from DNA isolation and sequencing to advanced bioinformatic analyses for ready-to-publish data. Our experienced staff have extensive expertise working with a variety of environmental and human sample types, including stool, saliva, soil, water and more. We are here to help!
Featured Publications
| Featured | ||
| Title | Pre-analytical sample stabilization by different sampling devices for PCR-based COVID-19 diagnostics | |
| Journal | New Biotechnology. 25 September 2022. | |
| Authors | Melina Hardt, Esther Föderl, Höbenreich,Stephanie Freydl, Antonio Kouros, Martina Loibner, Kurt Zatloukal | |
| Title | Evaluating Diagnostic Accuracy of Saliva Sampling Methods for Severe Acute Respiratory Syndrome Coronavirus 2 Reveals Differential Sensitivity and Association with Viral Load |
| Journal | The Journal of Molecular Diagnostics. October 2021. |
| Authors | Pieter Mestdagh, Michel Gillard, Sharonjit K.Dhillon, Jean-Paul Pirnay, Jeroen Poels, Jan Hellemans, Veronik Hutse, Celine Vermeiren, Maxime Boutier,Veerle De Wever, Patrick Soentjens, Sarah Djebara, Hugues Malonne, Emmanuel André, Marc Arbyn, John Smeraglia, Jo Vandesompele |
| Title | Exposure to traffic-related air pollution and bacterial diversity in the lower respiratory tract of children |
| Journal | PLoS ONE 16(6). June 24, 2021. |
| Authors | Christine Niemeier-Walsh, Patrick H. Ryan, Jaroslaw Meller, Nicholas J. Ollberding, Atin Adhikari, Tiina Reponen |
| Title | Entamoeba gingivalis: epidemiology, genetic diversity and association with oral microbiota signatures in North Eastern Tanzania |
| Journal | Journal of Oral Microbiology . 19 May 2021. |
| Authors | Christen Rune Stensvold, Michelle Nielsen, Vito Baraka, Rolf Lood, Kurt Fuursted & Henrik Vedel Nielsen |
| Title | The mycobiomes and bacteriomes of sputum, saliva, and home dust |
| Journal | INDOOR AIR. Volume 31, Issue 2. March 2021. |
| Authors | Christine Niemeier-Walsh, Patrick H. Ryan, Jarek Meller, Nicholas J. Ollberding, Atin Adhikari, Reshmi Indugula, Tiina Reponen |
| Title | A Novel Homozygous Missense Mutation of WEE2 Causes Female Infertility Characterized by Fertilization Failure |
| Journal | Research. 2021. |
| Authors | Wenwen Liu, Guijun Yan, Ningyuan Zhang, Na Kong, Min Wu, Junshun Fang |
| Title | Oral microbiota and dental caries data from monozygotic and dizygotic twin children |
| Journal | Scientific data. 13 October 2020. |
| Authors | Yelda Kasimoglu, Mine Koruyucu, Sinem Birant, Ilker Karacan, Nursen Topcuoglu, Elif Bahar Tuna, Koray Gencay & Figen Seymen |
| Title | Salivary microRNA 155, 146a/b and 203: A pilot study for potentially non-invasive diagnostic biomarkers of periodontitis and diabetes mellitus |
| Journal | PLOS ONE. August 2020. |
| Authors | Natheer H. Al-Rawi, Farah Al-Marzooq, Ahmed S. Al-Nuaimi, Mahmood Y. Hachim, Rifat Hamoudi |
| Title | Rapid and label-free isolation of small extracellular vesicles from biofluids utilizing a novel insulator based dielectrophoretic device |
| Journal | Lab on a Chip. 2019. |
| Authors | Leilei Shi, Damaris Kuhnell, Vishnupriya J. Borra, Scott M. Langevin, Takahisa Nakamuracef and Leyla Esfandiari |
| Title | The association of novel polymorphisms with stress fracture injury in Elite Athletes: Further insights from the SFEA cohort |
| Journal | ORIGINAL RESEARCH. JUNE 01, 2018. |
| Authors | Ian Varley, David C. Hughes, Julie P. Greeves, Trent Stellingwerff, Craig Ranson, William D. Fraser, Craig Sale |
| Title | Diversity and signature of small RNA in different bodily fluids using next generation sequencing |
| Journal | BMC Genomics. May 2018. |
| Authors | Mohamed El-Mogy, Bernard Lam, Taha A. Haj-Ahmad, Shannon McGowan, Darrick Yu, Lucas Nosal, Nezar Rghei, Pam Roberts & Yousef Haj-Ahmad |
| Title | Loneliness in Relation to Depression: The Moderating Influence of a Polymorphism of the Brain Derived Neurotrophic Factor Gene on Self-efficacy and Coping Strategies |
| Journal | Front. Psychol. 19 July 2017. |
| Authors | Marc Bedard*, Robbie Woods, Carly Crump and Hymie Anisman |
Technical Documentation
- Viral Inactivation Using Norgen’s Total Nucleic Acid Preservative and Norgen’s Saliva RNA Preservative
- Inactivation of Coronavirus in Saliva Samples
- The Influence of Saliva Preservatives and DNA Isolation Methods for Oral Microbiota Metagenomics
- Viability Of Microorganisms in Saliva & Stool Collection and Transportation Preservatives
- Comparison of Norgen’s Saliva/Swab RNA Purification Kit to a Magnetic Bead-Based Competitor's Kit for Viral RNA Extraction
- Comparison of Norgen’s Saliva/Swab RNA Purification Kit to Zymo’s MagBinding Quick-Viral DNA/RNA kit for Viral RNA Extraction
- Saliva Donor Flowchart
Technical Documentation
- Comparative Study of DNA Isolated from Saliva Preserved in Norgen’s Preservative Using Norgen’s Saliva DNA Isolation Kit Versus Qiagen’s QIAamp DNA Blood Mini Kit
- Stability of DNA Stored in Norgen’s Saliva DNA Preservative for 52 Months at Room Temperature
- The Use of Saliva Swabs in Norgen’s Saliva DNA Collection, Preservation and Isolation Kit
- Linearity of DNA Isolated from Increasing Volumes of Preserved Saliva Using Norgen’s Saliva DNA Isolation Kit
- The Effect of Elution Volume on DNA Quantity and Quality Using Norgen’s Saliva DNA Isolation Kit
- In‐Depth Analysis of the Microbial Diversity in Saliva Samples Preserved at Ambient Temperature for over 6 Years
- Rapid and qPCR ready DNA isolation from environmental and human bodily fluid samples using Magnetic bead system
- The Influence of Saliva Preservatives and DNA Isolation Methods for Oral Microbiota Metagenomics
- High Efficiency Real-Time PCR from Saliva DNA
- Inhibitory Effect of Norgen’s Saliva DNA Preservative on the Growth of Bacteria and Yeast
- Isolation of High Quality Saliva DNA from Variable Donor Saliva Samples
- Long Term Stability of DNA Stored in Norgen’s Saliva DNA Preservative
- Sequencing Analysis of Saliva DNA Isolated using Norgen’s Saliva DNA Collection, Preservation and Isolation Kit
- A Comparative Study Between Two Saliva DNA Preservation Systems, and Two Column-Based Saliva DNA Purification Methods
- Comparison of Norgen’s Saliva DNA Collection, Preservation and Isolation Kit to a Leading Competitor’s Kit
- DNA Isolation from Saliva Preserved with Norgen’s Saliva DNA Collection and Preservation Device using Qiagen’s QIAamp DNA Blood
- Efficient Restriction Enzyme Digestion of Saliva DNA isolated using Norgen’s Saliva DNA Collection, Preservation & Isolation Kit
- Stability of DNA Stored in Norgen’s Saliva DNA Preservative for 52 Months at Room Temperature
- The Range of DNA Yield with Norgen's Saliva DNA Collection, Preservation, and Isolation Kit
- Sex Determination from Saliva DNA Isolated using Norgen's Saliva DNA Collection, Preservation, and Isolation Kit
- The Range of DNA Yield and Purity with Norgen's Saliva DNA Collection, Preservation, and Isolation Kit
- Column-Based Isolation of DNA from Preserved Saliva Samples Offers High Quality, Consistent, and Rapid DNA Isolation
- The Use of Saliva Swabs in Norgen’s Saliva DNA Collection, Preservation and Isolation Kit
- Linearity of DNA Isolated from Increasing Volumes of Preserved Saliva Using Norgen’s Saliva DNA Isolation Kit
- The Effect of Elution Volume on DNA Quantity and Quality Using Norgen’s Saliva DNA Isolation Kit
- Comparison of DNA Isolated from Saliva using Norgen’s Preservative and Saliva DNA Isolation Kit Vs Competitor's
- Stability of DNA Stored in Norgen’s Saliva DNA Preservative at 55°C
- Determination of the DNA Molecular Weight (MW) from different Norgen Columns and Isolation Methods
- In‐Depth Analysis of the Microbial Diversity in Saliva Samples Preserved at Ambient Temperature for over 6 Years - PAG 2016
- Viability Of Microorganisms in Saliva & Stool Collection and Transportation Preservatives
- Rapid and qPCR ready DNA isolation from environmental and human bodily fluid samples using Magnetic bead system
- The Influence of Saliva Preservatives and DNA Isolation Methods for Oral Microbiota Metagenomics
Related Videos
Saliva/Swab Automated RNA Purification Tutorial
Saliva DNA Isolation Tutorial - Automated Magnetic Bead System (Cat. RU62900)
Contact the Experts
Get in touch with Norgen's on site product specialists for more information or a direct quote for the product you are interested in.